SHANGHAI, March 6, 2026 /PRNewswire/ — CARsgen Therapeutics Holdings Limited (Stock Code: 2171.HK), a company focused on developing innovative CAR T-cell therapies, has announced its 2025 Annual Results.
Business Highlights

- Cash and cash equivalents were around RMB1,123 million as of December 31, 2025. Cash and cash equivalents at the end of 2026 are expected to be not less than RMB1,000 million. The net loss for the year ended December 31, 2025 was RMB103 million, representing a decrease of approximately 87% compared to the year ended December 31, 2024. In light of operational factors such as the changes in operating cash flow, we expect to have adequate cash into the 2030.
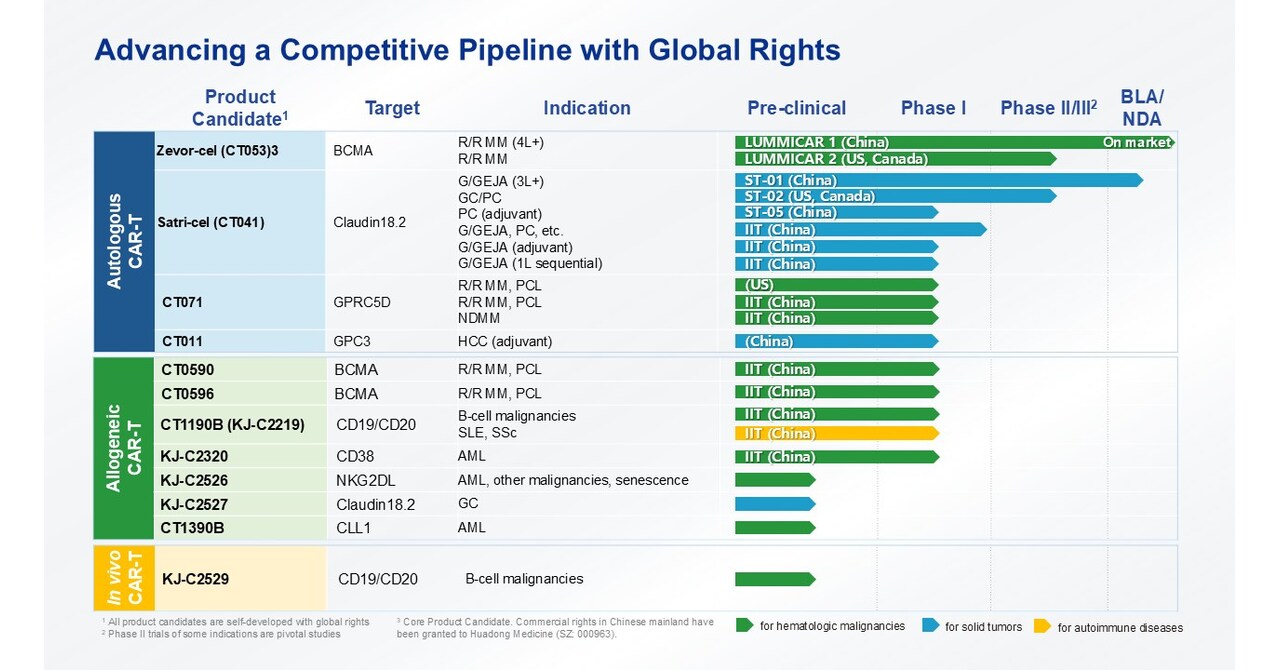
- During 2025, CARsgen has received a total of 218 confirmed orders of zevor-cel from its commercialization partner Huadong Medicine. In December 2025, zevor-cel has been included in China’s Commercial Health Insurance Innovative Drug Catalogue (2025).
- The Center for Drug Evaluation (CDE) of the National Medical Products Administration (NMPA) of China has accepted the New Drug Application (NDA) and granted Priority Review for satri-cel. The results of satri-cel confirmatory Phase II trial in China have been simultaneously published in The Lancet and at the 2025 ASCO Annual Meeting.
- Multiple allogeneic CAR-T products are in development, covering treatment areas such as hematologic malignancies, solid tumors, and autoimmune diseases.
- CARsgen independently developed the lentiviral-based CARvivo™ platform for creating in vivo CAR T-cell products.
- CARsgen entered into strategic cooperation agreements with a key platform enterprise in Jinshan District, Shanghai to establish an advanced commercial manufacturing base for CAR T-cell products.

Source link











Leave a Reply