Highlights:
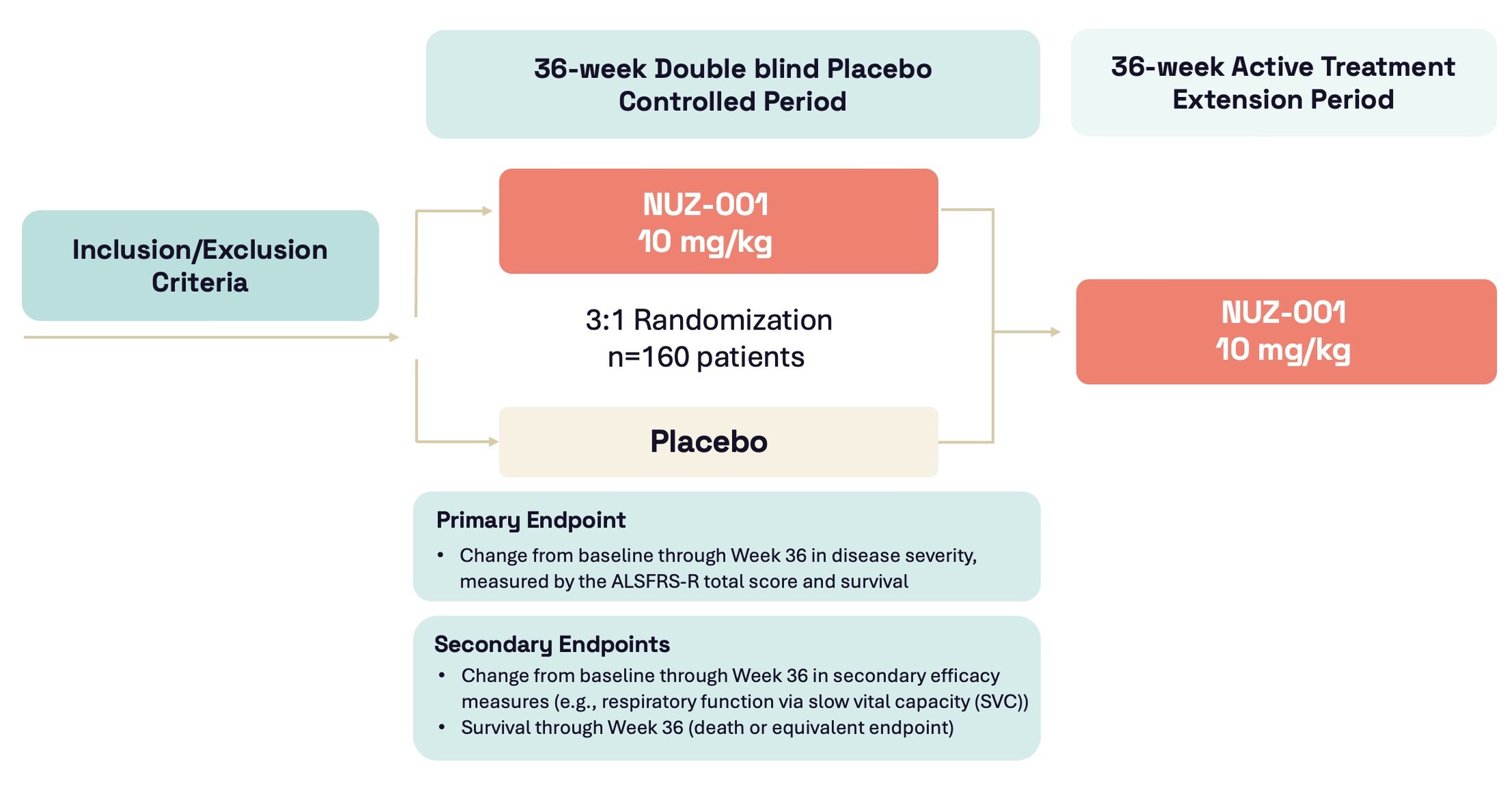
- First participant enrolled and dosed with NUZ-001 in Regimen I of the HEALEY ALS Platform Trial evaluating NUZ-001 for the treatment of ALS
- Approximately 160 participants with ALS will be enrolled in a 36-week randomised, double-blind, placebo-controlled adaptive Phase 2/3 clinical trial in leading ALS clinical centres across the United States
- Follows a successful Phase 1 clinical program in a small study population (n=12) in people living with ALS, which showed encouraging preliminary signals of efficacy and NUZ-001 was safe and well-tolerated
- Study is expected to complete enrolment in H2 CY2026

Source link













Leave a Reply