“AI Conductor transforms clinical development from a series of disconnected manual tasks into a synchronized, intelligent process,” said Raviv Pryluk, PhD, CEO and Co-founder of PhaseV. “Our AI doesn’t work in isolation. It draws from a wide knowledge base to deliver insights, enforce document consistency across the full trial, and ensure every study is submission-ready from the start.”
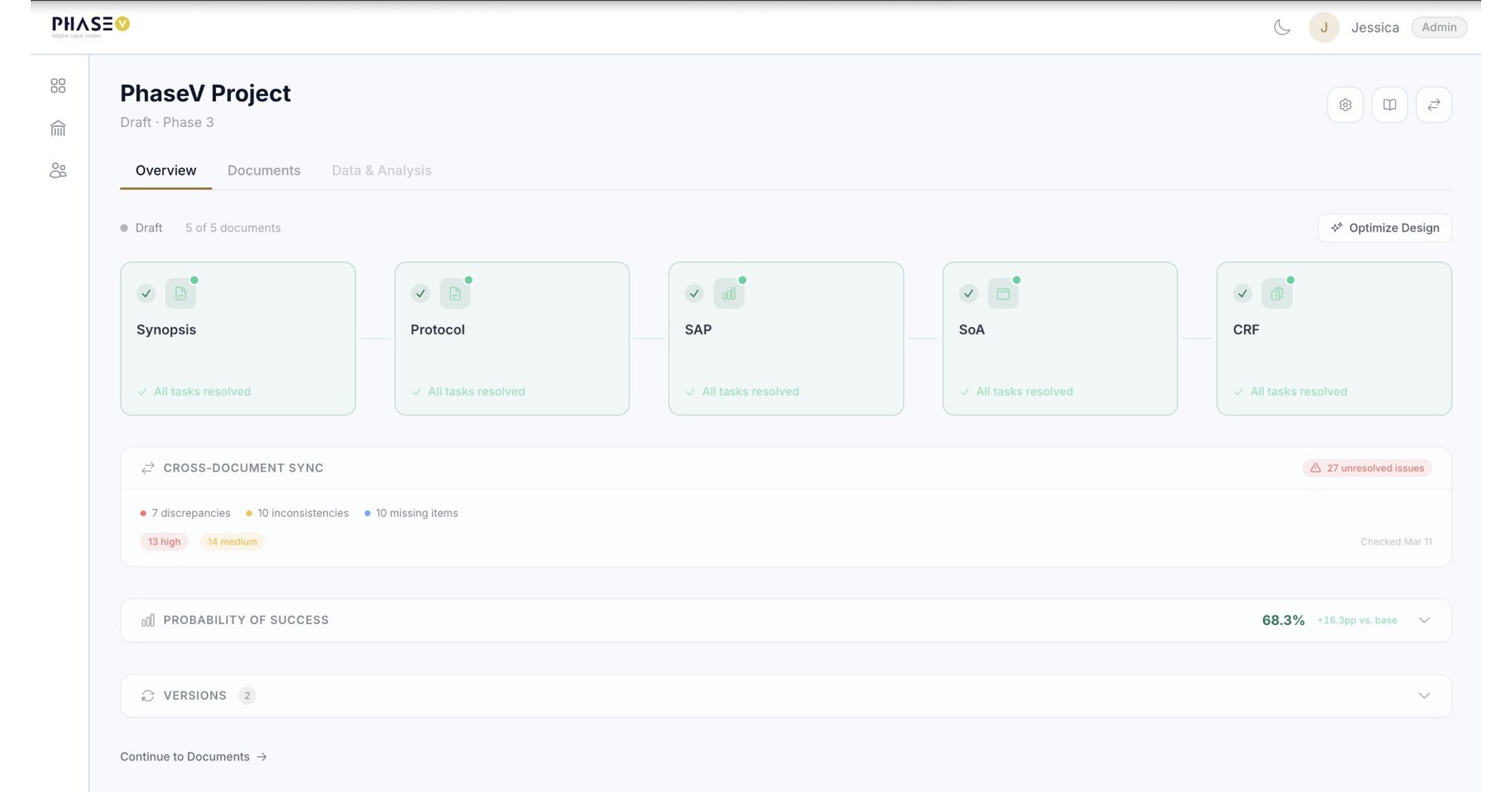
AI Conductor provides a highly configurable, enterprise-grade workspace that serves as a single source of truth across teams. While trial design traditionally involves fragmented conversations and numerous document iterations, PhaseV’s generative AI platform actively drafts study components and offers real-time optimizations. The platform grounds projects in regulatory logic by connecting to internal standard operating procedures (SOPs), historical documents and templates. This creates a shared space where teams can collaborate through granular permissions, inline commenting, and easy version comparisons while maintaining an audit-ready history of every change.
AI Conductor serves as a vital layer within PhaseV’s vertical AI platform, orchestrating the ClinOps, Trials, Responses, and Portfolio Optimizers. While these Optimizers help sponsors streamline trial design, maximize site selection and monitoring, identify target patient subgroups, and drive smarter clinical decision-making, AI Conductor guides users through several core capabilities across the trial lifecycle:
FINN Partners for PhaseV
[email protected] Photo – https://mma.prnewswire.com/media/2935893/PhaseV.jpg
Logo – https://mma.prnewswire.com/media/2267452/5868311/PhaseV_logo.jpg SOURCE PhaseV
- Pre-Trial Authoring & Collaboration: From Synopsis to AI-driven Protocol Authoring, the system ensures regulatory compliance at every step. It aligns Statistical Analysis Planning (SAP) with ICH E9 guidelines and generates CDASH-aligned Case Report Forms (CRFs) directly from visit schedules.
- Post-Data Integration & Processing: AI Conductor manages the full data pipeline through seamless EDC integration and automated data standardization. The platform also handles SDTM mapping and ADaM dataset generation including statistical code to produce publication-ready Tables, Listings, and Figures (TLFs) and Clinical Study Reports ready for FDA submission.
FINN Partners for PhaseV
[email protected] Photo – https://mma.prnewswire.com/media/2935893/PhaseV.jpg
Logo – https://mma.prnewswire.com/media/2267452/5868311/PhaseV_logo.jpg SOURCE PhaseV

Source link














Leave a Reply